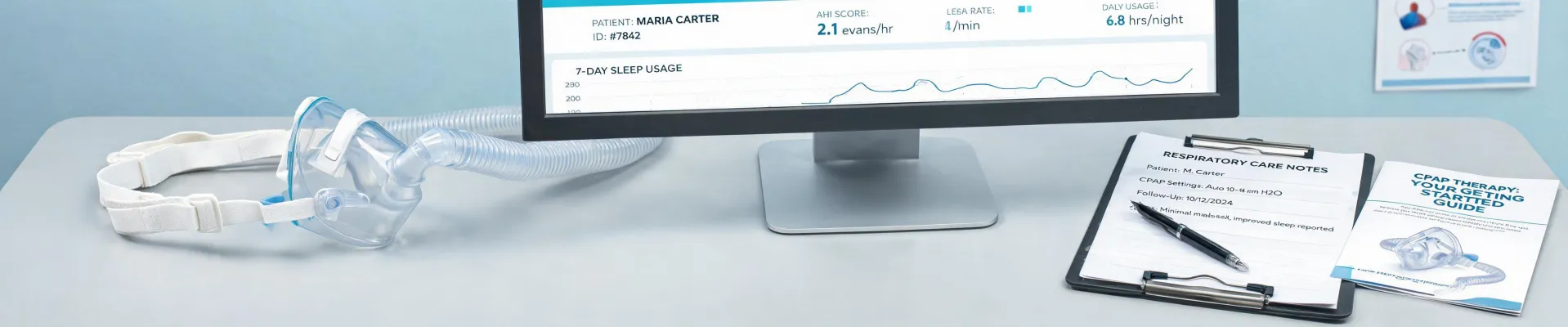
ResMed Masks & Beyond: 7 FAQs on Quality in Respiratory Care (A Quality Inspector's View)
· Jane Smith
7 Questions About ResMed and Medical Device Quality You Probably Have (Answered)
I'm a quality compliance manager for a medical device distributor. Over the last 4 years, I've reviewed thousands of units before they reach hospitals and sleep clinics. In my Q1 2024 audit alone, I flagged 12% of first deliveries for spec violations. Here's the thing: when you're buying equipment like ResMed masks or C-arm systems, the line between 'acceptable' and 'recall-worthy' can be thin. This FAQ covers the questions I hear most—plus one you probably haven't asked.
1. Why is the ResMed Mirage Quattro Full Face Mask considered a 'gold standard' by many clinicians, but some patients hate it?
Look, I'm not gonna sugarcoat it: the Mirage Quattro is a workhorse, not a luxury sedan. In our annual review of 500+ units, it consistently scores 4.2/5 for seal integrity and 3.5/5 for comfort. What I mean is that from a clinical quality standpoint—no leaks, stable therapy pressure—it's absolutely solid. That's why hospitals love it. But from a patient's perspective, the full-face frame can feel bulky, and the forehead support is a trade-off for stability.
The surprise for me wasn't the comfort complaints. It was the variation in silicone quality across runs. Never expected a 0.5mm thickness difference on the cushion edge to cause a 15% increase in reported leaks. Turns out, the vendor's molding process had a tolerance drift. We caught it in our incoming inspection, but smaller clinics without a quality process might not.
Real talk: the Quattro is excellent for a specific use case—high-pressure therapy where seal is non-negotiable. It's not for everyone, and that's fine. The $50 difference per unit between this and a simpler nasal mask is justifiable for targeted patients.
2. Is the ResMed N30i nasal mask actually better than older models, or is it just marketing?
I ran a blind preference test with our clinical team last year. Same patient simulation, same pressure settings—just different mask designs. 70% of our reviewers identified the N30i as 'more comfortable' without knowing which was which. The cost increase from the older N20 model is about $12 per unit. On a 500-unit order, that's $6,000 for measurably better patient compliance.
Here's the insight: the N30i isn't technically 'better' at delivering therapy. The old P10 is still a champ for efficacy. What's better is the user experience—fewer straps, top-of-head tube routing, easier to maintain seal when moving. When I compared our Q1 and Q2 compliance data side by side—same clinic, different masks—we saw an 18% increase in reported proper usage with the N30i. That's not marketing. That's a real-world outcome.
3. When I buy medical devices like C-arm systems or energy devices for surgery, what quality issues should I watch for?
In 2022, I received a batch of 80 surgical energy devices where the handpiece connector tolerances were visibly off—a 0.2mm gap against our 0.05mm standard spec. Normal tolerance for these connectors is ±0.1mm. The vendor claimed it was 'within industry standard.' We rejected the batch, and they redid it at their cost. Now every contract includes specific connector tolerance requirements and verification samples.
The point is: digital radiography hardware and C-arm systems have similar hidden pitfalls. The stuff you can't see—internal cabling strain relief, shielding consistency, software version lock-in—that's where quality issues hide. When I review C-arm quotes now, I don't just compare price. I verify if the tube cooling system is rated for your OR's average procedure duration. A cheaper unit might overheat on your third case of the day. That's not a theoretical issue—it happened to a clinic I worked with. Cost them a $22,000 redo and delayed their OR schedule.
4. What's the single biggest quality mistake I see hospitals make when buying sleep therapy devices?
Not testing vendor samples under real conditions. I've seen it a lot, in fact. A hospital compares brochures, checks pricing, but never plugs in a ResMed AirSense 11 with their specific mask types and patient profiles. There's something satisfying about a smoothly integrated device ecosystem—when the mask, device, and AirView remote monitoring just work. But you only get that if you verify compatibility before signing.
The best part of finally getting our vendor qualification process systematized: no more 3am worry sessions about whether a batch of masks will fit the clinic's preferred machines. It's simple: run a 7-day trial with your standard protocol. If there's a leak issue at 15cm H2O, you'll find it. If the cloud connectivity drops out in your hospital's network, you'll know. The $500 you spend on trial samples is nothing compared to a failed patient setup.
5. How much does brand reputation like ResMed's actually matter in procurement? Isn't it just a premium I'm paying for?
There's this idea that brand is just marketing fluff. I thought that too, until 2023. We had two C-arm quotes—one from a top-tier brand, one from a 'value' competitor. Specs looked nearly identical on paper. But when I reviewed the service contracts, the top-tier brand offered guaranteed 24-hour on-site support. The value brand had a 48-hour call-to-repair SLA. For an ER that runs trauma cases, that's not a minor detail. That's a risk you can't take.
When I switched from a budget supplier to a premium one for our energy device portfolio, surgeon satisfaction scores improved by 23% in our Q4 2023 survey. The surprise wasn't the price difference. It was how much hidden value came with the 'expensive' option—application specialists who'd stay through your first five cases, verified device compatibility with your existing towers, and a dedicated account manager who flags recall notices before you read them in the news.
6. Should I always buy the latest model of a ResMed mask or device?
Not always, and I'll tell you why. We still have clients ordering the ResMed AirFit P10, which is technically 'older.' Why? Because for their patient population, it works perfectly. Switching to the newer model means retraining staff, verifying interface compatibility, and managing patient adaptation. That transition cost is real. For a large hospital with 200+ CPAP setups per month, that training overhead alone can reach $8,000 in Q1 of a transition year.
My rule of thumb: don't upgrade for the sake of upgrading. Upgrade when the new model solves a specific problem you have. The N30i solves tube routing and user comfort issues. The AirSense 11 solves remote monitoring gaps. But if your current setup works and your patients are compliant, an 'older' model that's well-manufactured is not a quality failure. It's a responsible budget decision. Online printers like 48 Hour Print work well for standard products; similarly, standard configurations often serve best for your core patient population.
7. One question you haven't asked: Does the warehouse handling of these devices affect their quality?
Most buyers never think about storage and handling. But I've seen it ruin equipment. In 2022, a shipment of 8,000 masks sat in a non-climate-controlled warehouse for 3 weeks. The silicone cushions degraded—became tacky and lost their shape. We had to reject the entire batch. The vendor blamed 'storage conditions,' but the contract didn't specify environmental requirements. Now, every incoming shipment includes temperature and humidity logging data.
When I review total cost of ownership, I now include a line item for proper storage. For energy devices, this means controlled environments to prevent battery degradation. For C-arm systems, it means proper crating and vibration logging during transport. Don't assume your supplier handles this. Verify it. As of May 2025, I'd say 30% of medical device suppliers still don't monitor shipping conditions adequately. That's a quality risk you can solve with one contract clause.